A Cochrane review concluded that in early stage Hodgkin lymphoma (HL), combined modality therapy results in improved overall and disease free survival compared with either chemotherapy or radiation therapy (RT) alone.r In addition, secondary malignancy rates were lower with combined therapy when compared with RT alone. The number of cycles of doxorubicin, bleomycin, vinblastine, and dacarbazine (ABVD) required depends on the stage of disease. A large German randomised trial (HD10) demonstrated that the intensity of therapy can be reduced in early favourable disease without altering outcome.r Engert et al. showed that two cycles of ABVD with 20Gy of involved-field RT (IFRT) is equivalent to 4 cycles of ABVD with 30 Gy IFRT in early stage favourable disease, the previous standard of care.r
In patients with higher-risk disease (early unfavourable HL) progression free survival (PFS) and overall survival (OS) are inferior compared with early favourable disease.r The German Hodgkin Study Group has recently trialled (HD11) increasing the intensity of chemotherapy for the early unfavourable patients.r There was no benefit to giving 4 cycles of bleomycin, etoposide, doxorubicin, cyclophosphamide, vincristine, procarbazine, and prednisone (BEACOPP) baseline compared with 4 cycles of ABVD, when followed by IFRT.
In the HD-14 trialr, patients with early unfavourable disease were randomised to either 4 cycles of ABVD + 30 Gy RT or 2 cycles of ABVD + 2 cycles esc BEACOPP + 30 Gy RT. Although PFS improved with the 2+2 regimen, it did not impact OS. However, a recent Cochrane reviewr concluded that “this meta-analysis provides moderate- to high-quality evidence that adult patients between 16 and 60 years of age with early unfavourable and advanced stage HL benefit regarding OS and PFS from first-line chemotherapy including escalated BEACOPP".
Multiple trials have considered the omission of RT for early stage favourable disease.rr Radford et al., found only a modest improvement of 3 year PFS in favour of RT in an intention-to-treat analysis (95% vs 91% without RT), however, longer analysis is required to assess the late effects of RT. In a further study by the EORTC/LYSA/FIL H10 group, an increased number of events were noted in the non-RT arm in the early unfavourable risk group (PET directed after 2 cycles of ABVD) and the study was therefore closed early.r HD 16 trial patients with early stage favourable disease were randomised to either 2 cycles of ABVD + 20 Gy RT (CMT) or 2 ABVD with no RT in patients who are PET negative. Among 628 PET-2-negative, per-protocol-treated patients, 5-year PFS was 93.4% (95% CI, 90.4% to 96.5%) with CMT and 86.1% (95% CI, 81.4% to 90.9%) with ABVD (difference 7.3% [95% CI, 1.6% to 13.0%]; hazard ratio, 1.78 [95% CI, 1.02 to 3.12]). Five-year OS was 98.1% (95% CI, 96.5% to 99.8%) with CMT and 98.4% (95% CI, 96.5% to 100.0%) with ABVD. In conclusion, in PET-2-negative patients, RT cannot be omitted from CMT without clinically relevant loss of tumour control. The study also showed a poor PFS for patients who are Deauville 4 at the end of 2 cycles. (5-year PFS, 93.1% [95% CI, 90.7% to 95.5%] v 80.9% [95% CI, 72.2% to 89.7%]; P = .0011).r
The latest Cochrane review publishedr of PET-directed therapy for early stage HL concluded that “to date, no robust data on OS, response rate, treatment-related mortality (TRM), quality of life (QoL), or short- and long-term adverse events (AEs) are available". However, this systematic review found moderate-quality evidence that PFS was shorter in individuals with early-stage HL and a negative PET scan receiving chemotherapy only (PET-adapted therapy) than in those receiving additional RT (standard therapy). It is thus regarded that RT is important in early stage HL patients receiving ABVD (2-4 cycles).
For patients with an interim PET 2 positive the EORTC/LYSA/FIL H10 Trial demonstrated an improved efficacy for patients to be changed to esc BEACOPP after 2 cycles. In the trial of the 1,950 randomly assigned patients, 1,925 received an ePET—361 patients (18.8%) were positive. In ePET-positive patients, 5-year PFS improved from 77.4% for standard ABVD + Involved-nodal radiation therapy (INRT) to 90.6% for intensification to BEACOPPesc + INRT (hazard ratio [HR], 0.42; 95% CI, 0.23 to 0.74; P = 0.002).r
Efficacy
In patients with early stage HL, combined modality therapy using ABVD and IFRT results in CR rates of 96-100 %.rrrr Long term (7-12 years) freedom from treatment failure is 87-95% and OS 94-96%.
Figure 1: Freedom from progressionr
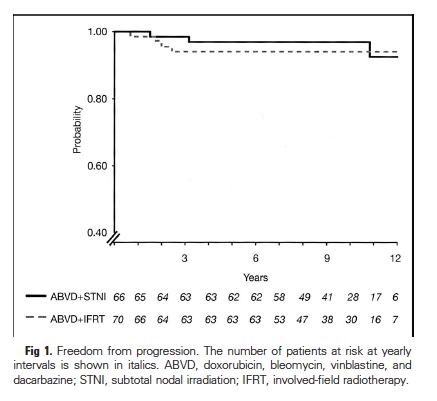
© Journal of Clinical Oncology 2004
Figure 2: Overall survivalr

© Journal of Clinical Oncology 2004
Toxicity
ABVD causes Grade 3-4 neutropenia in 30-34% of patients.rr Despite this, rates of significant infections are low (1-2%, Grade 3-4),rr Evens et al.r demonstrated that G-CSF support is not required to maintain near 100% dose intensity during ABVD treatment. In their study, 58% of 658 ABVD treatments were administered with an ANC <1.0 x 109/L without an increase in infection (incidence rate 0.44%) when compared with those given G-CSF (0.78%). Other Grade 3-4 early toxicities include alopecia 24-30%,rr nausea and vomiting 6-13%rr and pulmonary 6-10%rr Fertility is relatively well preserved after ABVD. 30-54% of people assigned male at birth experience transient oligoazoospermia, however, most fully recover.rr Transient amenorrhoea occurs in around one-third of people assigned female at birth of reproductive age (<45 years), but permanent amenorrhoea secondary to ABVD alone is uncommon.rr Furthermore, people assigned female at birth attempting pregnancy after ABVD had fertility comparable with case controls with a 12 month pregnancy rate of 70%.r Although second malignancies occur in patients post-ABVD (6% at 6-10 years follow up) rates of secondary myelodysplastic syndrome (MDS)/acute myeloid leukaemia (AML) are very low and do not appear to be significantly increased.rrr Other long term toxicities include bleomycin-induced lung injury (link to Pulmonary toxicity associated with anti-cancer drugs) and doxorubicin-related cardiac toxicity (link to Cardiac toxicity associated with anthracyclines).
In the most recent Cochrane reviewr, authors concluded that: “The risk of secondary AML/MDS is increased, but efficacy is improved among patients treated with intensified chemotherapy protocols. Treatment decisions must be tailored for individual patients. Consolidating radiotherapy is associated with an increased rate of secondary malignancies; therefore, it appears important to define which patients can safely be treated without RT after chemotherapy, both for early and advanced stages. For early stages, treatment optimisation methods such as use of fewer chemotherapy cycles and reduced field or reduced-dose radiotherapy did not appear to markedly affect efficacy or secondary malignancy risk. Due to the limited amount of long-term follow-up in this meta-analysis, further long-term investigations of late events are needed, particularly with respect to secondary solid tumours. Since many older studies have been included, possible improvement of RT techniques must be considered when interpreting these results.