The evidence supporting the use of this regimen comes from the TAX 324 trial, a randomised, multicentre open-label, phase III trial.r Between May 1999 and December 2003, a total of 501 patients with a WHO performance status of 0 or 1, were randomised to one of two arms:
Arm 1 (TPF): docetaxel 75mg/m2 and cisplatin 100 mg/m2 on day 1 followed by fluorouracil 1000 mg/m2/day from day 1 to day 4
Arm 2 (PF): cisplatin 100 mg/m2 on day 1 followed by fluorouracil 1000 mg/m2/day from day 1 to day 5
The cycles were repeated every 3 weeks for 3 cycles. All patients who did not have progressive disease were to receive chemoradiation therapy (carboplatin with concurrent radiation therapy).
The primary endpoint was overall survival. Patients with tumours of the nasopharynx and nasal/paranasal cavities were excluded from this study.
Efficacy
After a median follow up of 42 months, treatment with TPF resulted in a 30% reduction in the risk of death (HR=0.70; p=0.006). The median survival was 71 months in the TPF group and 30 months in the PF group (p=0.006). There was better locoregional control in the TPF group than in the PF group (p=0.04), but the incidence of distant metastases in the two groups did not differ significantly (p=0.14).r
Kaplan-Meier estimates of (A) Overall Survival and (B) Progression-free survivalr
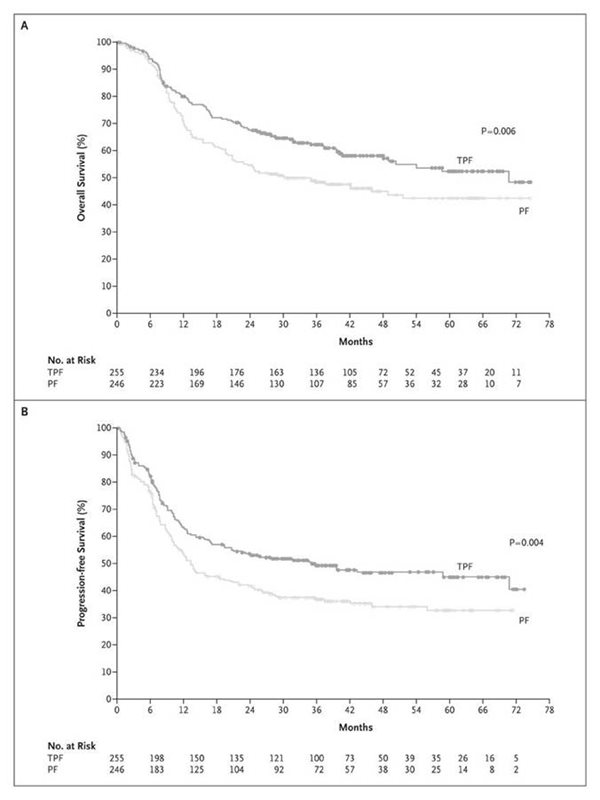
© New England Journal of Medicine 2007
Toxicityr
Rates of neutropenia and febrile neutropenia were higher in the TPF group and rates of nonhaematologic toxicity were similar in the 2 study groups.r

© New England Journal of Medicine 2007